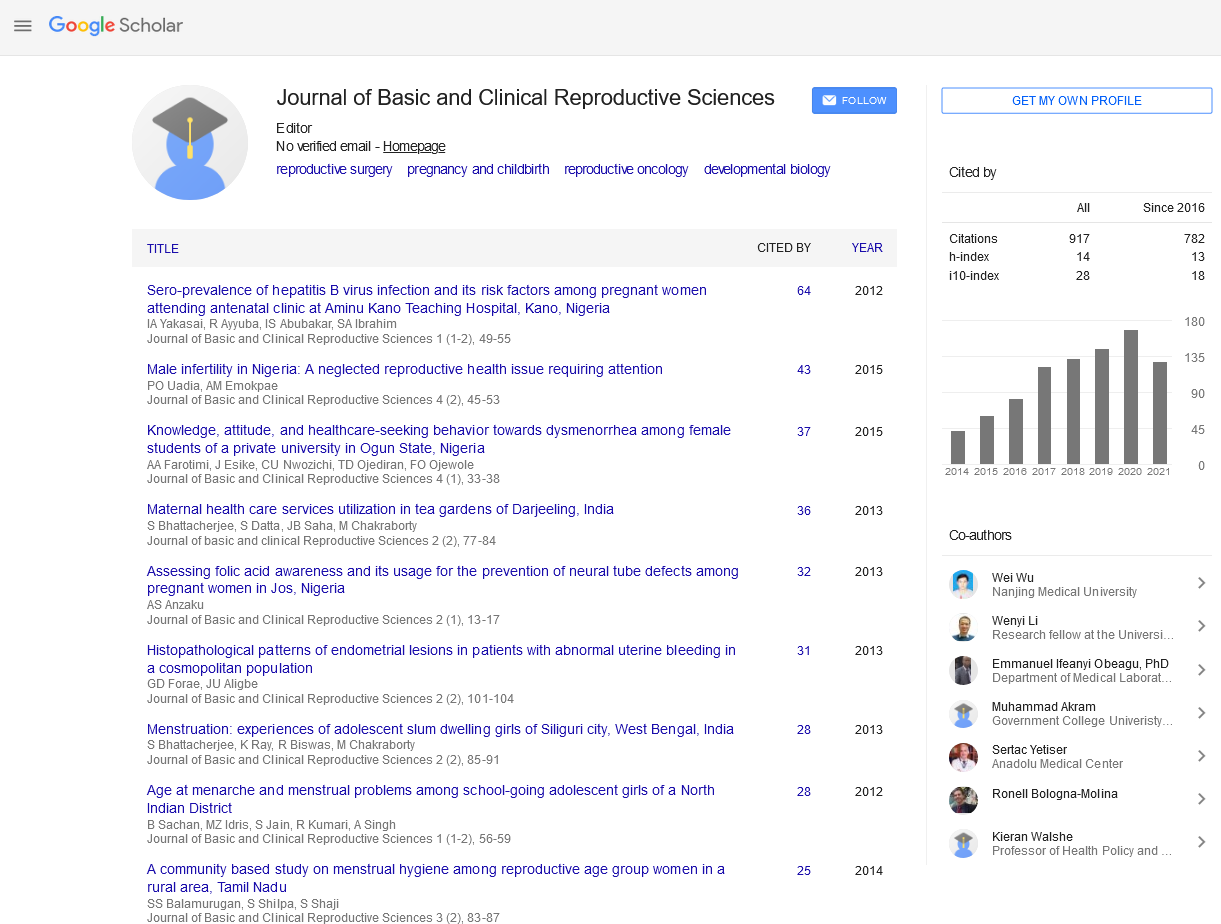
Short communication - Journal of Basic and Clinical Reproductive Sciences (2019) Volume 8, Issue 1
IUI or IVF: In Resource Crunch Countries.
Received: 20-Dec-2018 Accepted Date: Jan 23, 2019 ; Published: 01-Feb-2019
Citation: Bajaj B IUI or IVF: In Resource Crunch Countries
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com